Here’s our background as a medical device manufacturer,
how we provide value to society, and what we aim to achieve.
Our background
ORIGIN
JMS was established by a physician who
aimed to improve medical care settings.
-
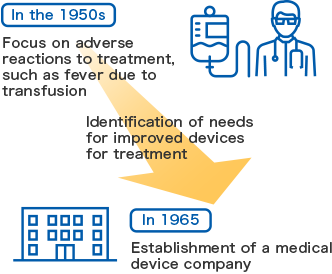
Around 1963, adverse reactions due to transfusions frequently occurred in medical care settings in Japan. Dr. Taro Tsuchiya, a PhD in medicine for his thesis on conserved blood, conducted unique investigational research into the adverse reactions, and determined that the cause was the reuse of transfusion sets. To provide solutions by promoting the use of disposable products in accordance with such practice in the United States, he established a company that markets disposable medical devices. That is how JMS started.
Our characteristics as a manufacturer
APPROACH
Always focus the clinical settings
and promote task-tackling R&D method.
-
-
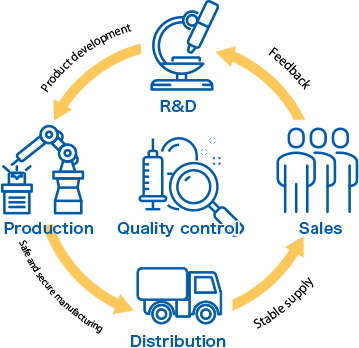
Established as a medical device manufacturer by a physician with a sense of mission, JMS not only develops and manufactures requested products but also supports actual medical care settings, providing unique solutions for healthcare professionals and patients. In developing products, JMS has established a process to identify the needs of medical care settings, discover hidden issues, and provide improvement. The findings obtained through development and other processes are given back to the clinical settings through activities such as seminars for healthcare professionals.
Our technologies
PRODUCTS
Address issues in clinical settings through our technologies to
appropriately control difficult-to-handle blood
and chemical solutions.
-
-
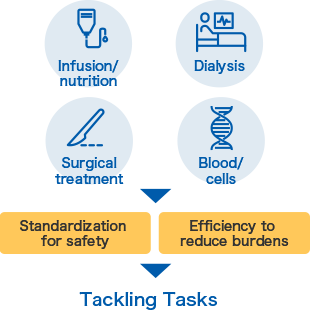
JMS provides a wide range of solutions, from the field of infusion/transfusion to the fields of infusion/nutrition, dialysis, surgical treatment, and blood/cells. One of our key focuses is standardization. This is a process of improvement to build a system to ensure safe medical activities in medical care settings where no errors should occur. Another important focus is efficiency. Our solutions to reduce burdens on staff in complex medical care settings contribute to the maintenance of high-quality medical care.
Overseas operations to provide our technologies
GLOBAL
Establish a system to provide safer and easier-to-use medical devices
in any country at any time.
-
-
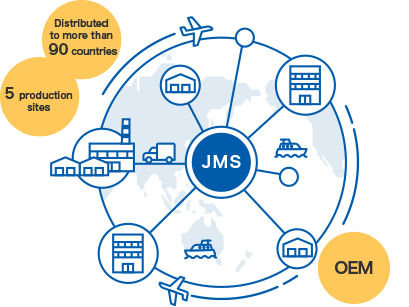
JMS has started overseas operations in the 1970s, and currently supplies products to more than 90 countries. JMS has production/sales bases mainly in southeast Asia where demand for medicine is growing. We have established a system to enable the distribution of medical devices needed on-site more speedily. Our “product development/production/supply according to on-site needs” remains the same irrespective of our areas of activity. We will continue to promote our global operations in the years to come to enable the JMS brand to permeate throughout the world.
JMS future vision
VISION
Focus on next-generation’s challenge;
focus on the world’s medical care’s challenge.








